AMA, AHA, ANA Send Trump Administration Letter Encouraging Transition

The Biden transition team needs full cooperation and all critical information regarding the COVID-19 coronavirus, he American Medical Association (AMA), American Hospital Association (AHA) and American Nurses Association (ANA) said in a November 2020 letter to the Trump administration.
- Read more about AMA, AHA, ANA Send Trump Administration Letter Encouraging Transition
- Add new comment
Telegenetic Counseling Bridges Geographic Barriers and Minimizes Distress

Our 2009–2014 study, Bridging Geographic Barriers: Remote Cancer Genetics Counseling for Rural Women, also known as the REACH Project (Risk Education and Assessment for Cancer Heredity), was the first randomized, noninferiority trial of telephone-based BRCA1 and BRCA2 counseling and testing that used a population-based traceback approach to identify and counsel both rural and urban women who were at increased risk for hereditary breast and ovarian cancer but had not received genetic counseling or testing.
- Read more about Telegenetic Counseling Bridges Geographic Barriers and Minimizes Distress
- Add new comment
FDA Warns Against Wearing Face Masks With Metal During MRI Exams

The U.S. Food and Drug Administration (FDA) issued an alert on December 7, 2020, informing patients and healthcare providers that patients may be injured if they wear face masks with metal parts and coatings during magnetic resonance imaging (MRI) exams. Metal parts (e.g., nose pieces, nanoparticles, or antimicrobial coatings that may contain metal such as silver or copper) may become hot and burn patients during MRIs.
Manage Cancer Treatment-Related Constipation With ONS Guidelines™

As many as 43%–58% of patients with cancer experience constipation related to their treatment, and the side effect is both distressing and potentially life threatening if severe enough. Constipation-related emergency department visits increased by 41.5% from 2006–2011, with older adults (aged 85 years or older) making up most of the visits. Estimates suggest that the cost of managing severe cancer-related constipation may range from $500 to more than $2,300 per person per month. Supporting patients who experience the side effect is critical for their well-being.
A New Year Offers Promise of Improvement

I don’t know how many times this year I’ve thought to myself or heard someone say, “When will 2020 be over?” The pandemic, social unrest, political polarization, and natural disasters have made 2020 a year in which many of us have wondered, “What else?”
Nursing Innovation Links Rural Facilities to Resources and Experts to Provide High-Quality Care Across the Country

When a patient comes to the doctor’s office with a generalized symptom such as an ongoing cough or chest heaviness, they are bound to have anxiety. In Wayne’s case, a patient with a lung mass that was identified unexpectedly, his anxiety was further compounded by feeling alone: his wife has dementia and his grown children live out of state. He also lives in a rural area and must drive a significant distance to get to a center for testing and diagnosis.
- Read more about Nursing Innovation Links Rural Facilities to Resources and Experts to Provide High-Quality Care Across the Country
- Add new comment
Pancreatic Cancer Risk Factors, Diagnosis, Treatment, Side Effects, and Survivorship Considerations

Pancreatic cancer is the ninth most common cancer in the United States, accounting for 3% of all cancers but causing 7% of cancer-related deaths, which equates to about 57,500 diagnoses and 47,050 deaths each year. The average person’s risk for pancreatic cancer is about 1 in 64.
- Read more about Pancreatic Cancer Risk Factors, Diagnosis, Treatment, Side Effects, and Survivorship Considerations
- Add new comment
FDA Approves Pralsetinib for RET-Altered Thyroid Cancers

On December 1, 2020, the U.S. Food and Drug Administration (FDA) approved pralsetinib for patients aged 12 years and older with advanced or metastatic RET-mutant medullary thyroid cancer who require systemic therapy or RET fusion-positive therapy and who are refractory to radioactive iodine (if radioactive iodine is appropriate).
NKT Cells May Be the Next Wave of CAR Therapy
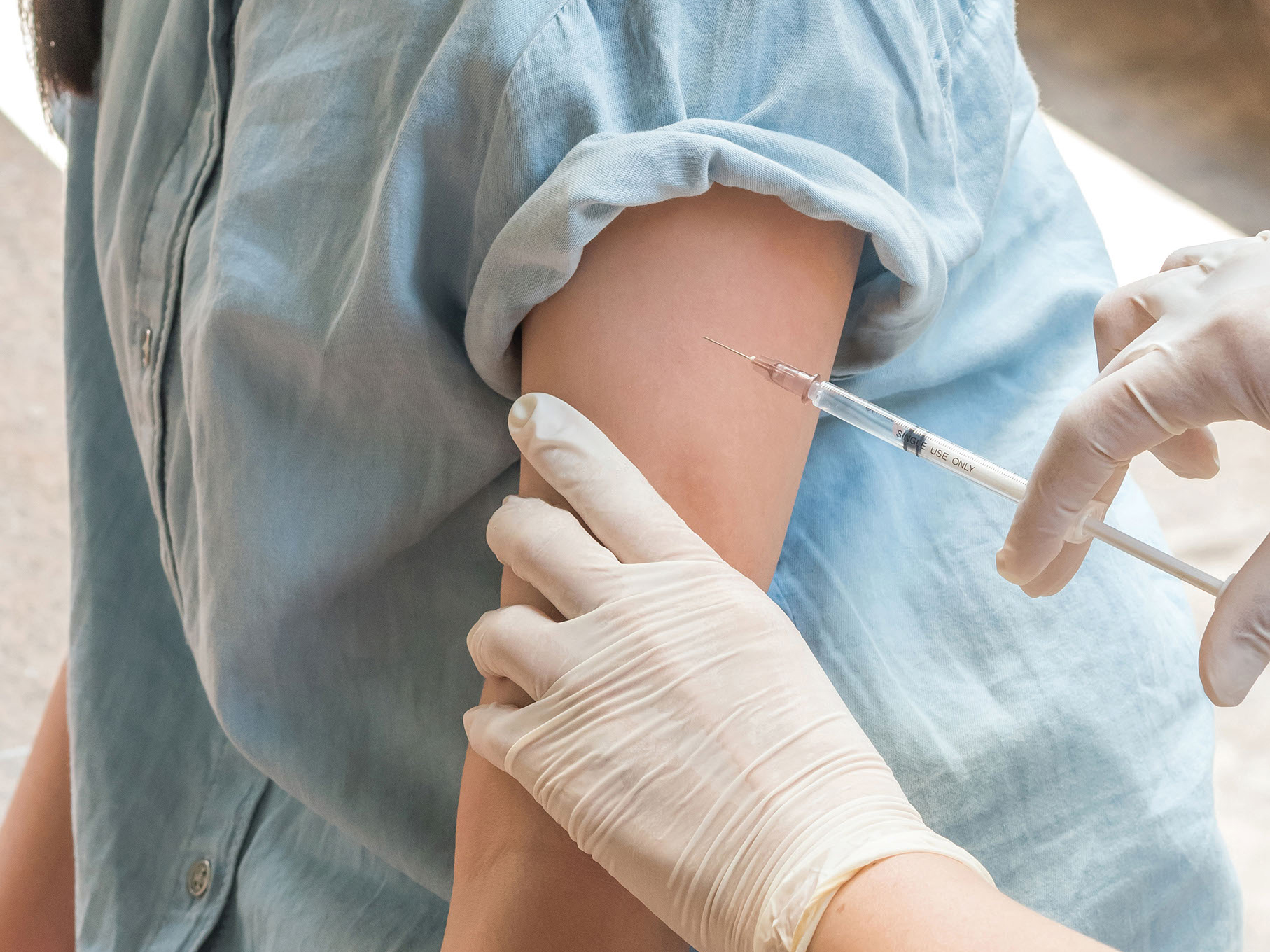
When genetically modified with a chimeric antigen receptor (CAR), natural killer T (NKT) cells eliminated 50% of metastases in a patient with heavily pretreated, relapsed or refractory metastatic neuroblastoma, according to interim findings from an ongoing study that were published in Nature Medicine.
FDA Offers Guidance to Enhance Diversity in Clinical Trials

The COVID-19 coronavirus continues to smother the United States, and nationwide efforts to flatten the curve aren’t lowering cases or preventing deaths. U.S. Food and Drug Administration (FDA) Commissioner Stephen M. Hahn, MD, an oncologist by training and profession, addressed the actions needed to combat COVID-19. One in particular is ensuring that clinical trials accurately reflect diverse populations.





